Healthcare analysts explain why they are bullish on Allergan plc Ordinary Shares (NYSE:AGN) and Zafgen Inc (NASDAQ:ZFGN). Although many investors are worried that new U.S. treasury regulations threaten the planned Pfizer-Allergan merger, one analyst believes the stock is strong enough to withstand such an event. On another note, positive phase 3 data from Zafgen makes one analyst bullish on an upcoming FDA meeting discussing the results.
Allergan plc Ordinary Shares
Nomura analyst Shibani Malhotra of Nomura commented on Allergan today following the U.S. Treasury’s new regulations intended to limit inversions and “earnings stripping.” The analyst explains how this will affect the $160 billion merger with Pfizer, as the latter is relocating its headquarters to Ireland, where AGN is located, in order to avoid a higher U.S tax rate. However, Pfizer will maintain most of its operations in the U.S.
The analyst is mainly concerned with certain points in the notice, which discuss ownership percentages. When determining if an acquisition counts as an inversion, the treasury stated that “today’s action excludes stock of the foreign company attributable to assets acquired from an American company within three years prior to the signing date of the latest acquisition.” This is particularly worrisome due to AGN’s acquisitions in the past 3 years, which would constitute Pfizer’s stake in the company at over 60%, qualifying the merger as an inversion and “reducing the economic benefits of such a deal.”
Unlike the previous ‘7874 statue, the new regulations state that the IRS does not have to “demonstrate intent” that AGN planned to cheat the tax system. Rather, the IRS may disqualify the “stock issued in transactions within the last 3 years.” However, the analyst points out that “under current rules, the AGN/PFE merger is technically not an inversion, as post-merger, Pfizer shareholders are expected to own ~56% of the combined entity.” Furthermore, “action would be needed from Congress,” in order to enforce the new statue, which according to the analyst “is unlikely in an election year.” She continues, “We would therefore expect significant push back from various stakeholders to this somewhat arbitrary change in the ‘7874 inversion rule.”
The analyst notes that lack of commentary from both companies make her analysts preliminary. Despite the new regulations and their possible implications to disqualify the merger, the analyst believes in the strength of Allergan’s stock. She explains, “Most important, we believe that the current stock price does not reflect the standalone value of Allergan and we view AGN shares as compelling even absent a deal with Pfizer.” She continues, “In our view, on a standalone basis Allergan offers some of the best, most durable assets in the sector, it is run by a shareholder focused management team with strong operational abilities and has a solid mid-to-late stage pipeline that could deliver significant upside to our estimates.”
The analyst reiterates a Buy rating on the stock with a $350 price target.
According to TipRanks, Shibani Malhotra has a 45% success rate recommending stocks with an average loss of (4.2)% per recommendation. Out of the 10 analysts who have rated the stock in the past 3 months, all are bullish. The average 12-month price target for the stock is $350, marking a 50% upside from current levels.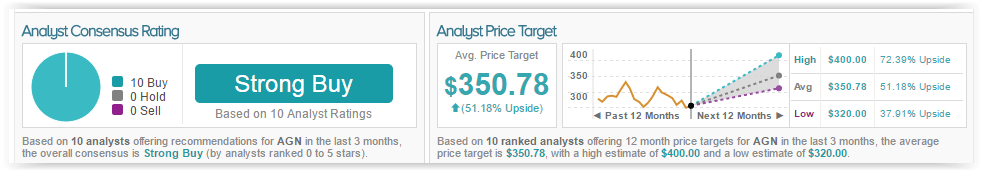
Zafgen Inc
Analyst Christopher James of FBR provided his insights on Zafgen after the company released positive phas 3 results from its best PWS trial with beloranib, used to treat Prader-Willi syndrome (PWS). PWS is a genetic condition characterized by unrealistic feelings of starvation, driving the patient to excessively eat and gain weight, known as hyperphagia which causes dangerous levels of obesity.
The results indicated that beloranib demonstrated a statistically significant LDL-C reduction (artery clogging cholesterol) “reduction in total body weight… compared to placebo” as well as reduced hyperphagia-related behaviors. The analyst views these results as positive, stating, “We believe these improvements in secondary endpoints further underscore the benefits of beloranib for a desperate population living with significant co-morbidities.”
Following the positive results, the company plans to meet with the FDA to review the data from its various studies on beloranib. However, James notes that the FDA placed a full clinical hold on beloranib in December 2015 following two patient deaths resulting from its use. The analyst will be watching for updates from the discussions related to the clinical hold status. Despite this setback, the analyst notes that “Overall, we believe the data further demonstrate a positive risk/reward profile for beloranib in patients with PWS, who lack an effective PWS therapy.”
The analyst reiterates an Outperform rating on the company with a $20 price target.
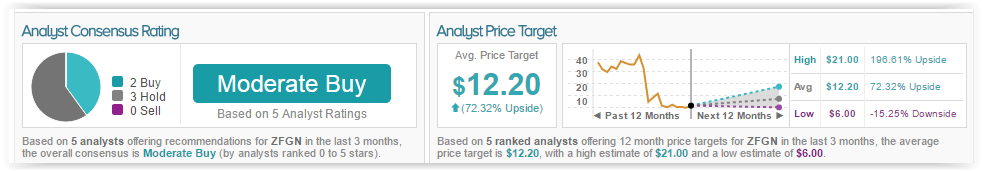
Christopher James has a 41% success rate recommending stocks with an average loss of (7.8%) per recommendation. According to TipRanks, out of the 5 analysts who have rated the company in the past 3 months, 2 are bullish and 3 remain on the sidelines. The average 12-month price target for the stock is $12.20, marking a 73% upside from current levels.