Johnson & Johnson is slated to report first-quarter 2021 earnings on April 20 before the market opens. Over the past six months, shares of the medical device company have gained 12.9% and are currently trading at $162.69. A strong set of numbers could be the catalyst for a big move upward, so let’s take a closer look at what analysts on the Street are expecting.
Earnings Preview
Johnson & Johnson (JNJ) is expected to report EPS of $2.34 on revenues of $22 billion. Meanwhile, the Earnings Whisper number stands at $2.41 per share. Notably, the Whisper number is the Street’s unofficial view on earnings.
Prior Period Results
In the prior quarter, the company reported adjusted earnings of $1.86, down 1.1% year-over-year. That said, this figure beat the consensus estimate of $1.82. Meanwhile, reported sales grew 8.3% to $22.48 billion and surpassed analysts’ expectations of $21.67 billion.
In the first quarter of 2021, Johnson & Johnson is expected to report EPS of $2.34 compared to the $2.30 reported in the same quarter last year. (See Johnson & Johnson stock analysis on TipRanks)
Factors To Look For
The company’s Pharma segment has been a top revenue generator over the past few quarters, with key products including Darzalex, Imbruvica, Stelara, Tremfya, and Erleada likely to have driven sales for the first quarter as well.
Furthermore, the Medical Devices segment, which reflected the impact of the market recovery in the fourth quarter of last year after being hit hard by the COVID-19 pandemic, is likely to have recorded gains in 1Q. However, the resurgence of the virus in the US and Europe might have disrupted surgical procedures to some extent, leading to subdued recovery trends.
The Consumer Health unit is likely to have benefited from increased demand for oral care products due to the pandemic. However, lower demand for skin health/beauty and international baby care products might have been an offsetting factor in the first quarter.
Recent Developments
In recent months, Johnson & Johnson was awarded Emergency Use Authorization (EUA) for its COVID-19 vaccine in the US by the Food and Drug Administration (FDA) and received Interim Order authorization in Canada. Furthermore, the company was also granted Conditional Marketing Authorization (CMA) in the European Union.
Additionally, the company also received the FDA’s approval for its PONVORY (ponesimod) therapy as a first-of-its-kind oral treatment for adults with relapsing forms of multiple sclerosis (MS).
In March, the World Health Organization (WHO) issued Emergency Use Listing (EUL) for Johnson & Johnson’s single-shot COVID-19 vaccine. The world’s largest healthcare company designed the vaccine to prevent COVID-19 in individuals 18 years of age and older.
However, recently, the US Centers for Disease Control and the Food and Drug Administration cited six reports of patients developing blood clots after receiving Johnson & Johnson’s COVID-19 vaccine and recommended a pause of its use until a decision is reached.
Analyst Recommendations
On April 13, Goldman Sachs analyst Terence Flynn reiterated a Buy rating on the stock following the FDA and CDC’s joint statement, which indicated a pause on the usage of the company’s COVID-19 vaccine pending further review of rare cases of blood clots.
Flynn awaits details regarding the risk/benefit profile of the J&J vaccine. However, he noted that a numerical imbalance existed in blood clots reported with the vaccine in the Phase 3 trial, while as per the FDA document, the data at that time was not sufficient to establish a fundamental relationship between these events and the vaccine.
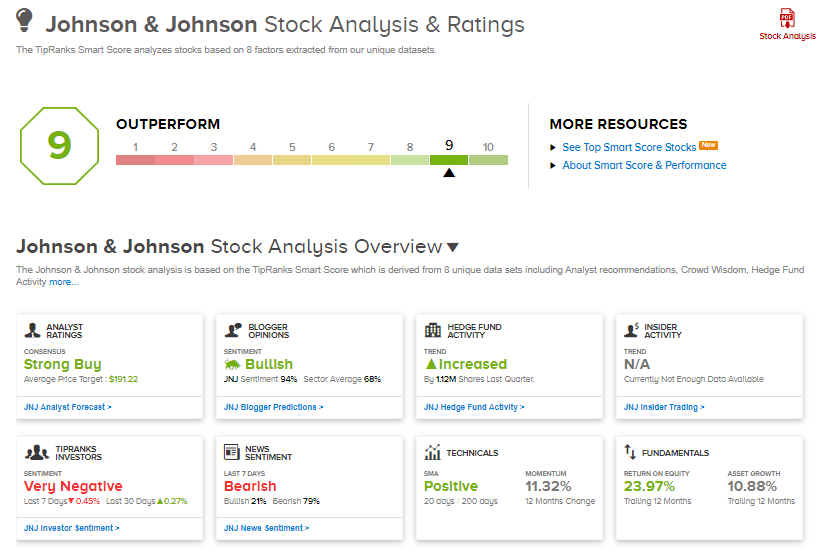
The consensus rating among analysts is a Strong Buy based on 9 Buys versus 1 Hold. The average analyst price target stands at $191.22 and implies upside potential of almost 17.5% to current levels.
Furthermore, Johnson & Johnson scores a 9 out of 10 from TipRanks’ Smart Score rating system, indicating that the stock has strong potential to outperform market expectations.
Related News:
Goldman’s 1Q Results Exceed Expectations As Revenues Outperform; Shares Gain
JPMorgan’s 1Q Results Beat Estimates On Strong Revenues
Wells Fargo Jumps 5.5% After A Blowout Quarter, Fee Revenue Escalates









