Biopharmaceutical company Ocugen, Inc. (OCGN) announced that the U.S. Food and Drug Administration (FDA) has accepted its Investigational New Drug Application (NDA) to initiate a first-in-human clinical trial of OCU400 (AAV-NR2E3).
The modifier gene therapy candidate is designed for the treatment of retinitis pigmentosa resulting from genetic mutations, which were found in NR2E3 and Rhodopsin. The therapy aims to treat multiple retinal diseases with a single product.
Shares of the company closed 6.3% lower on Thursday.
CEO Comments
Encouraged by the FDA approval, Ocugen CEO Shankar Musunuri said, “We are delighted to advance OCU400 into clinical trials, which exemplifies our goal of offering new options to people with genetic diseases where none currently exist.”
“We’re collaborating with leading centers in eye care and have been vital partners to getting our trial launched and receive patients. With this final decision by the FDA, we are embarking on a new pathway of care through this innovative gene therapy,” Musunuri added.
Prior Approvals
Between 2019 and 2020, the FDA granted four orphan drug disease designations to OCU400 for the treatment of four different gene mutation-related retinal degenerative diseases. Additionally, in 2021, the therapy received broad orphan medicinal product designation from the European Medicines Agency (EMA) for the treatment of both retinitis pigmentosa (RP) and Leber congenital amaurosis (LCA).
Wall Street’s Take
Recently, Noble Financial analyst Robert LeBoyer maintained a Buy rating and a price target of $15 (165.5% upside potential) on the stock.
The rest of the Street is cautiously optimistic about the stock, with a Moderate Buy consensus rating based on 2 Buys and 2 Holds. The average Ocugen price target of $8.75 implies 54.87% upside potential. Shares have lost 15.6% over the past six months.
Risk Analysis
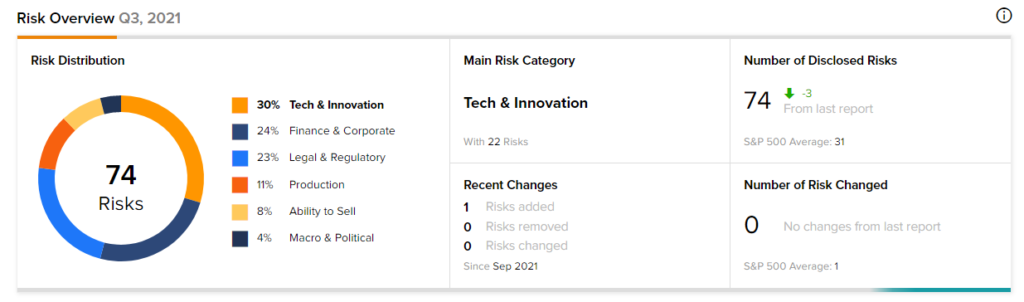
According to the new TipRanks Risk Factors tool, Ocugen stock is at risk mainly from three factors: Tech and Innovation, Finance and Corporate, and Legal and Regulatory, which contribute 30%, 24%, and 23%, respectively to the total 74 risks identified for the stock.
Related News:
GameStop Posts Wider-than-Feared Q3 Loss; Shares Drop After-Hours
Pfizer & BioNTech Provide Results on Omicron Vaccine Efficacy
Tilray Acquires Breckenridge Distillery; Shares Gain









