Junshi Biosciences Co Ltd said on Tuesday that it is gearing up to initiate clinical trials to test its experimental COVID-19 antibodies together with Eli Lilly & Co (LLY), in the U.S. and China by the second quarter of the year.
Eli Lilly, earlier this month announced its partnership with Junshi Biosciences, a China-based biopharmaceutical company specializing in the discovery and development of novel therapies. Since the coronavirus outbreak, Junshi Biosciences has created multiple neutralizing antibodies to combat COVID-19.
Junshi Biosciences also published in the science journal Nature, efficacy data results from preclinical studies that tested two of the experimental monoclonal antibodies, CA1 and CB6, in rhesus monkeys. The results showed that while both antibodies demonstrated “substantial” neutralization activity in vitro against SARS-CoV-2, CB6 exhibited “superior” neutralizing activities.
“The fact that CB6 can protect rhesus monkeys from COVID-19 infection suggests a potential for prophylactic use in humans,” said Daniel Skovronsky, Lilly’s chief scientific officer and president of Lilly Research Laboratories. “We are moving as quickly as possible to test such a protective effect in patients at risk for COVID-19.”
As part of the collaboration, Lilly will receive an exclusive license to conduct clinical development, manufacturing and distribution of products outside of Greater China. Junshi Biosciences will maintain all rights in Greater China.
Shares in Eli Lilly have been on a winning streak since March 23, advancing 24% to $147.56 as of early U.S. trading on Tuesday.
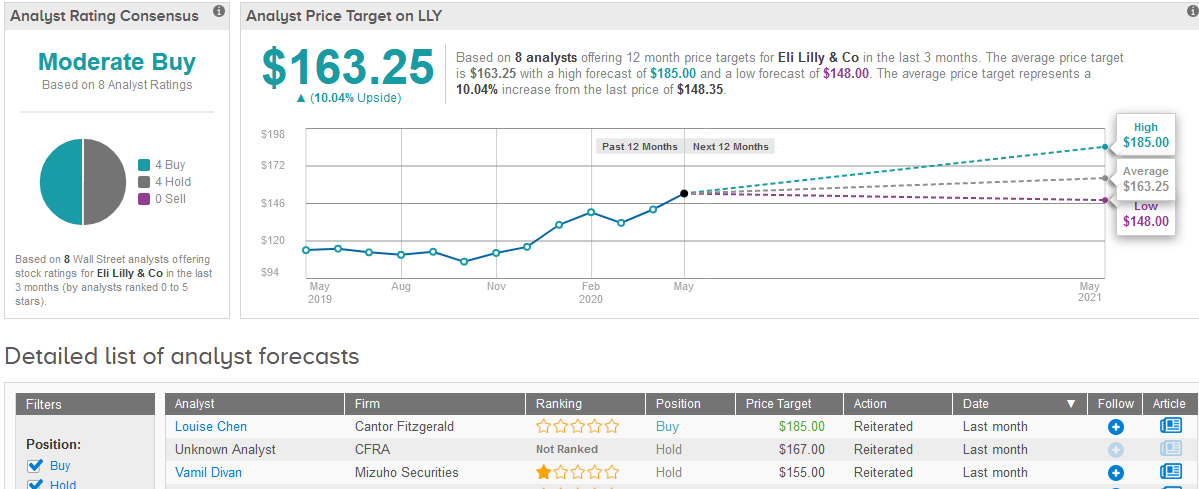
Wall Street analysts are cautiously optimistic about Lilly’s stock outlook divided evenly between 4 Buy ratings and 4 Hold ratings, which add up to a Moderate Buy consensus. The $163.25 average price target implies 11% upside potential of 5.2% in the coming 12 months. (See Eli Lilly’s stock analysis on TipRanks).
Related News:
Merck Joins Race For Covid-19 Vaccine; Shares Rise 4.4% In Pre-Market Trading
Novavax Begins Human Testing For Covid-19 Vaccine, Expects Results In July
Gilead’s Remdesivir Most ‘Beneficial’ In Covid-19 Patients Who Need Extra Oxygen, Study Shows