Medigen Vaccine Biologics (MVC) and Dynavax Technologies (DVAX) have announced that MVC has obtained a Taiwan government subsidy for successfully initiating a Phase 1 clinical trial in Taiwan.
The first participant in MVC’s Phase 1 clinical trial was dosed with MVC’s COVID-19 vaccine combined with Dynavax’s CpG 1018 adjuvant at National Taiwan University Hospital in early October. The subsidy will be released at agreed upon milestones in the total amount of up to NT$ 472 million (US$ 16.4 million).
The grant received by MVC was earmarked by the Taiwan government for purposes of research and development of a locally produced COVID-19 vaccine.
Shares in Dynavax are rising 7% in Tuesday’s pre-market trading.
MVC’s Phase 1 study is an open-label, single-center, dose-escalation study intended to assess the safety and immunogenicity of the SARS-CoV-2 recombinant spike protein S-2P at three dose levels adjuvanted with CpG 1018 plus alum, in 45 healthy subjects 20 to 50 years of age.
The vaccination schedule consists of two doses for each study participant, administered via intramuscular (IM) injection 28 days apart, on Day 1 and Day 29.
“The pre-clinical study results demonstrated that the combination of our S-2P vaccine candidate and CpG 1018 plus alum provided safety and immunogenicity sufficient to advance to Phase 1 development,” said Charles Chen, CEO of MVC. “We are pleased with the pre-clinical data and the potential to demonstrate clinical benefit.”
An adjuvant is a pharmacological or immunological agent that modifies the effect of other agents. Adjuvants are added to a vaccine to boost the immune response to produce more antibodies and longer-lasting immunity, thus minimizing the dose of antigen needed.
Dynavax says that its CpG 1018 adjuvant provides a well- developed technology and a significant safety database. This could potentially accelerate the development and large-scale manufacturing of a COVID-19 vaccine. (See DVAX stock analysis on TipRanks)
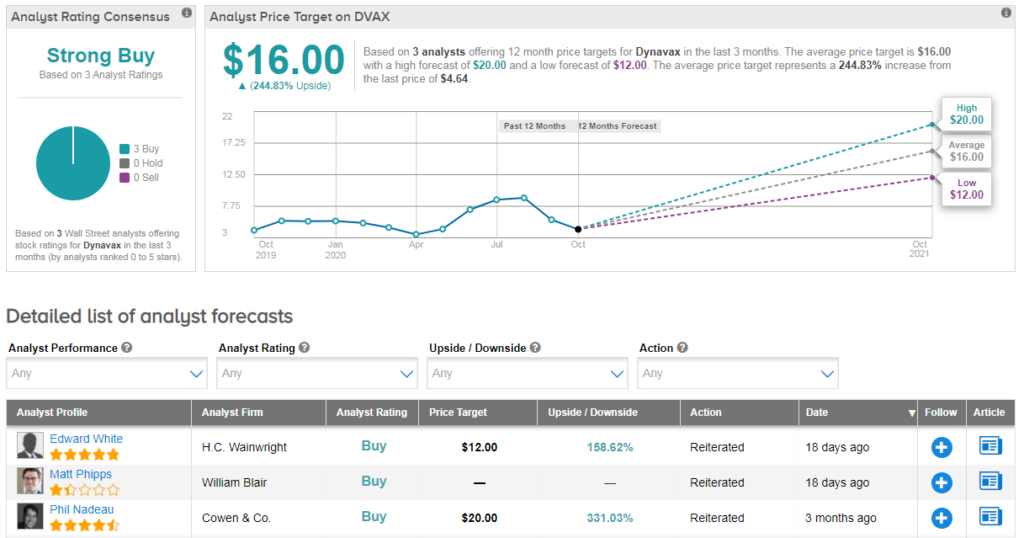
Shares in Dynavax are down 19% year-to-date, but the stock scores a bullish Strong Buy consensus from the Street. That’s with three recent buy ratings, and no hold or sell ratings. Meanwhile the average analyst price target stands at $16, indicating significant upside potential from current levels.
Dynavax has partnerships with five different companies developing COVID-19 vaccines that utilize Dynavax’s CpG 1018 adjuvant, notes HC Wainwright analyst Edward White. He has a buy rating on the stock and $12 price target, and estimates CpG 1018 adjuvant sales for COVID-19 vaccines of $25M in 2022 growing to $100M in 2028.
Related News:
PerkinElmer Raises 3Q Sales Outlook Fueled By COVID-19 Testing Demand
J&J Halts Covid-19 Vaccine Trial Due To ‘Unexplained Illness’
Pfizer, BioNTech Initiate Rolling Canada Submission For Covid-19 Vaccine