Altimmune, a clinical-stage biopharmaceutical company, and Switzerland-based Lonza, a global partner to the pharmaceutical, biotech, and nutrition markets, extended their AdCOVID manufacturing collaboration.
Per the terms of the extended agreement, clinical and commercial production of AdCOVID, Altimmune’s (ALT) single-dose intranasal vaccine candidate for COVID-19, will be supported by Lonza with a committed manufacturing suite at the company’s facility near Houston, Texas.
Altimmune VP of Product Development Dr. Vyjayanthi Krishnan said, “Manufacturing capacity for COVID-19 vaccines has been severely constrained, and this limitation has presented considerable challenges for vaccine developers.”
“By expanding our Lonza collaboration and commissioning our own dedicated manufacturing suite, we are building extra capacity and redundancy into our manufacturing to support potential late-stage clinical trials with AdCOVID and potential future commercial supply,” Krishnan added.
Based on clinical experience with its vaccine platform technology, Altimmune expects AdCOVID to provide immunity of up to a year or more with a single dose, with it having the capability for a favorable tolerability profile. (See Altimmune stock analysis on TipRanks)
Altimmune CEO Vipin K. Garg said, “We recently commenced our AdCOVID Phase 1 clinical trial and anticipate having a data readout in the second quarter of 2021.”
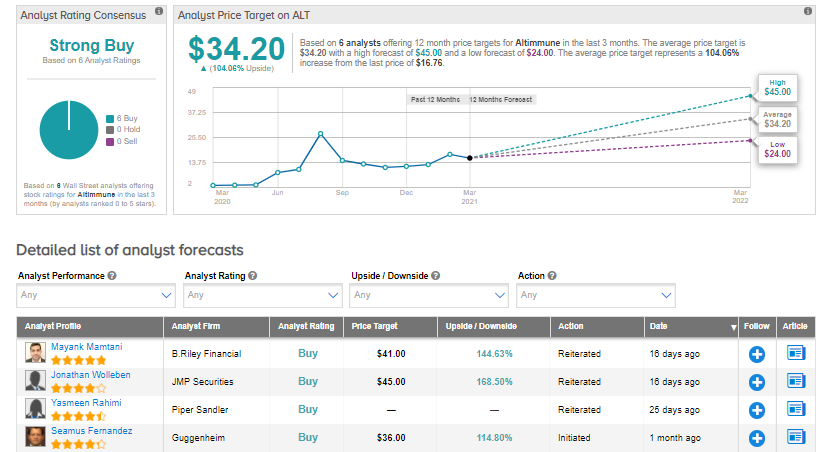
On Feb. 26, B. Riley Financial’s Mayank Mamtani reiterated a Buy rating on the stock with a price target of $41 (144.6% upside potential), as the analyst is “encouraged by the breakaway potential of ALT’s pipeline to be realized in a data-rich 2Q.”
Shares have rallied almost 32% so far this year, while Wall Street analysts are still bullish about the stock. The Strong Buy consensus rating boasts 6 unanimous Buys. Looking ahead, the average analyst price target stands at $34.20, putting the upside potential at 104% over the next 12 months.
Related News:
Cardinal Health Inks $1B Deal To Sell Cordis Business
Sanofi, Translate Bio Kick Off Human COVID-19 Vaccine Trial; Shares Gain 5%
Novavax’s COVID-19 Vaccine Is 96.4% Effective In UK Trial; Shares Pop 23%









