Clinical-stage specialty pharmaceutical company Zynerba Pharmaceuticals (ZYNE) has received guidance from the U.S. Food and Drug Administration (FDA) for a Phase 3 trial of Zygel to treat Fragile X syndrome (FXS).
FXS is a rare genetic disability that causes inherited intellectual disability as well as autism spectrum disorders.
In the Phase 3 trial, called RECONNECT, the efficacy and safety profile of Zygel will be evaluated in children and adolescents with FXS. Zygel, which is a gel that delivers CBD (non-euphoric cannabinoid) through the skin, targets the behavioral effects of FXS.
Zynerba Chairman and CEO Armando Anido said, “Following productive discussions and alignment with the FDA, we believe we have a clear path forward for Zygel in Fragile X syndrome. We are excited to advance Zygel into the RECONNECT trial, a pivotal, multi-national, confirmatory Phase 3 trial in patients with FXS in the third quarter of 2021.” (See Zynerba stock analysis on TipRanks)
Anido added, “If the results are positive, Zygel could become the first FDA approved treatment option for the significant unmet medical need that affects patients with FXS and their families.”
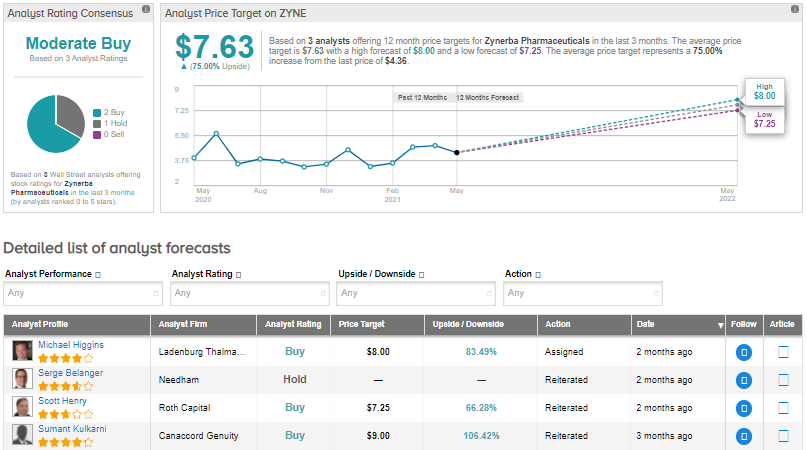
Recently, Roth Capital analyst Scott Henry reiterated a Buy rating on the stock but lowered the price target to $7.25 (66.3% upside potential) from $8.
Commenting on Zynerba’s Q4 results as “within expectations,” Henry also noted the sufficient cash available to the company, which will last until 1H 2024.
Consensus on the Street is that Zynerba Pharmaceuticals is a Moderate Buy based on 2 Buys and 1 Hold. The average analyst price target of $7.63 implies 75% upside potential. That’s after the stock has seen an approximate 17.5% gain over the past six months.
Related News:
Where Does Roku Stand Ahead Of Its 1Q Earnings Release?
PayPal Earnings Preview: Here’s What To Expect
Verizon To Sell Media Business To Apollo Management For $5B But Will Retain 10% Stake









