Regeneron Pharmaceuticals, Inc (REGN) announced on Tuesday that it has won a $450 million contract with the U.S government to manufacture and supply REGN-COV2, its investigational dual antibody cocktail for the prevention and treatment of COVID-19.
The stock rose 3.6% to $650 in early afternoon trading. The agreement, which was signed with the U.S. Department of Defense and the Biomedical Advanced Research and Development Authority (BARDA) will fund continued manufacturing so that the therapy could be made available immediately in the U.S. if clinical trials are successful and the U.S. Food and Drug Administration (FDA) grants Emergency Use Authorization (EUA).
The agreement, which is part of Operation Warp Speed, a U.S. government program that seeks to begin supplying millions of doses of a safe, effective vaccine for COVID-19 in 2021, covers a fixed number of bulk lots intended to be completed in the fall of 2020, the company said.
“We made the decision early on to begin large-scale manufacturing at our own risk in order to ensure that product would be available immediately if our clinical trials prove successful and an Emergency Use Authorization is granted,” said Regeneron CEO Leonard S. Schleifer. “This manufacturing and supply agreement could help REGN-COV2 reach many people quickly, hopefully helping to change the course of this deadly and still-raging pandemic.”
REGN-COV2 is currently in two Phase 2/3 clinical trials for the treatment of COVID-19 and in a Phase 3 trial for the prevention of COVID-19 infection. Regeneron said that the ongoing REGN-COV2 clinical program is evaluating multiple dosages and will help establish the exact number of potential treatment doses or prevention doses available from these lots in total. Initial doses may be ready as early as end of summer, the company added.
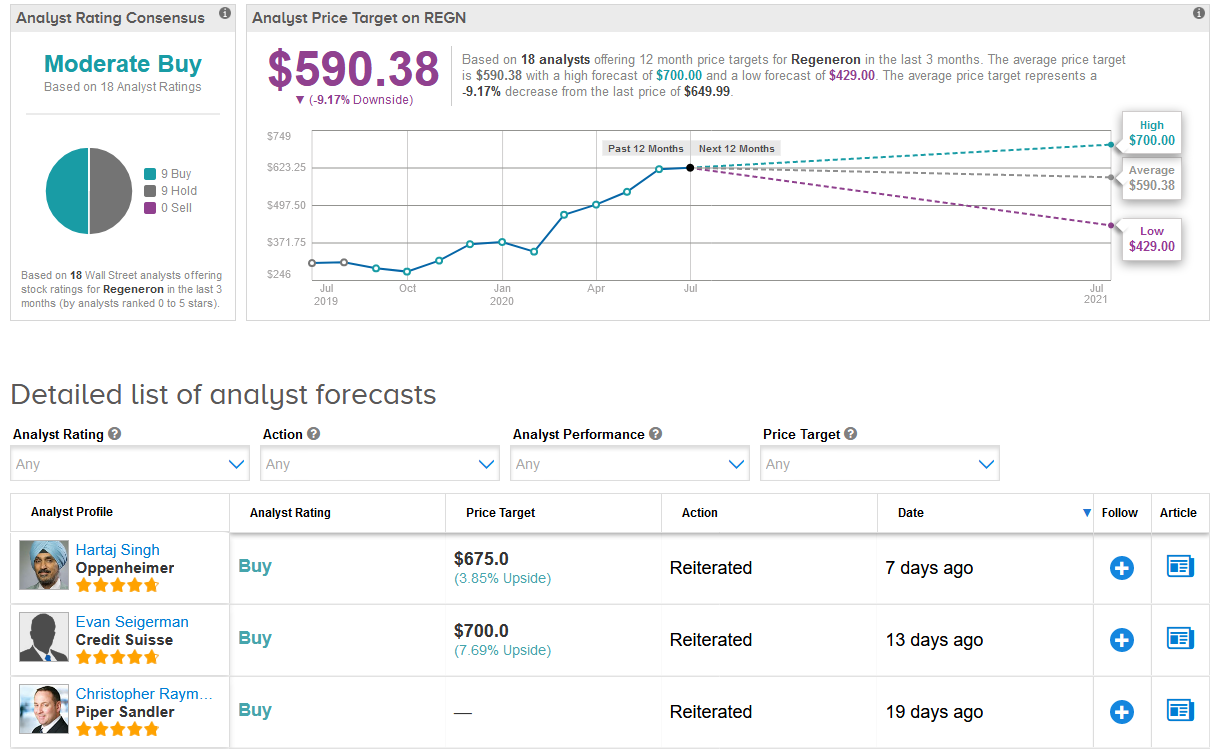
In the run-up to finding a treatment against COVID-19, shares in Regeneron have leaped 73% so far this year. In view of the stock’s recent rally, the $590.38 average price target now indicates 9% downside potential in the coming 12 months.
Oppenheimer analyst Hartaj Singh recently raised the stock’s price target to $675 from $625 and maintained a Buy rating, saying that the company is one of his top biotech names, characterized by ongoing product launches, commercial execution, OPEX control, and a best-in-breed pipeline.
“We believe this quality biotech name deserves a higher multiple,” Singh wrote in a note to investors. “Given COVID-19 effects on sales and clinical trial follow-through lacking in FY20 visibility, we caution that further performance could be accompanied by greater volatility. We view sustained weakness as a buying opportunity.”
Overall, the rest of the Street is cautiously optimistic on the stock. The Moderate Buy consensus breaks down evenly between 9 Buys and 9 Holds. (See Regeneron stock analysis on TipRanks).
Related News:
Novavax Spikes 42% Pre-Market On $1.6B U.S. Funding For Covid-19 Candidate
Corvus Shoots Up 115% On Start Of Novel Immunotherapy Study In Covid-19 Patients
Regeneron Up 5% In Pre-Market On Start Of Late-Stage Covid-19 Antibody Trial