Diagnostic solutions provider Quidel Corp. (QDEL) received an amended use authorization (EUA) from the U.S. Food and Drug Administration (FDA) for its Sofia Q device.
Sofia Q is a Fluorescent Immunoassay Analyzer (FIA) from the company’s Sofia product portfolio that provides the same accuracy in SARS Antigen testing as Sofia and Sofia 2 and delivers automated results in 15 minutes.
Quidel President and CEO Douglas Bryant said, “Sofia Q is our latest powerful diagnostic instrument designed to democratize access to the many benefits of our Sofia SARS rapid antigen tests and, ultimately, our full portfolio of Sofia tests for influenza, RSV, Strep, and other conditions.” (See Quidel stock analysis on TipRanks)
Bryant added, “We designed Sofia Q to be very affordably priced and conducive to widespread adoption across the ever-expanding global point-of-care and telemedicine marketplace.”
Additionally, users can pair the Sofia Q device with a Sofia Q mobile application that guides users and uses in-house AI modeling to interpret test results. After initial marketing to professional and point of care channels, Quidel plans to offer the new device to telemedicine and home markets.
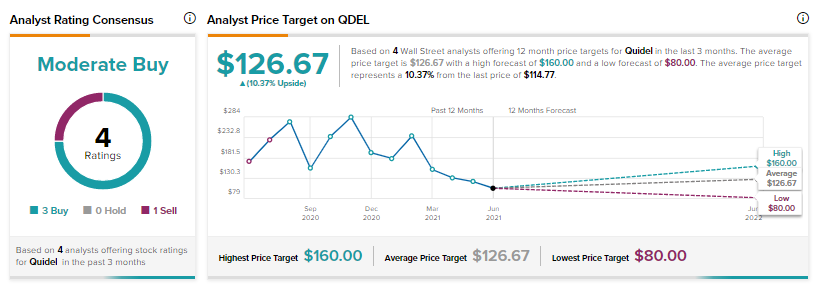
On June 1, Raymond James analyst Andrew Cooper reiterated a Buy rating on the stock and a $160 price target (39.4% upside potential).
Cooper believes despite a demand decline, “an instrumented option like Sofia will win in a true clinical setting versus visually read options. Multiyear contracts should translate to a stable customer base as well (versus clinicians likely moving away from visually read tests).”
Based on 3 Buys and 1 Sell, consensus among analysts is a Moderate Buy. The QDEL average analyst price target of $126.67 implies 10.4% upside potential.
That’s after shares have seen a 39.7% price correction so far this year.
Related News:
United States Steel Sells Transtar for $640M, Shares Rise 3%
Southwest Airlines Foresees Revenue Growth; Orders 34 More Boeing 737 MAX Aircraft
CareDx Acquires Transplant Hero, Shares Rise









