Pfizer Inc. announced a multi-year agreement with Gilead Sciences, Inc. to manufacture and supply Gilead’s investigational antiviral remdesivir.
Financial details weren’t disclosed. Under the terms of the agreement, Pfizer (PFE) will provide manufacturing services at its McPherson, Kansas facility to produce and supply remdesivir. With this deal, the drugmaker is joining a number of external manufacturing organizations as Gilead is under pressure to shore up supply of the investigational treatment for COVID-19.
Gilead (GILD) expects to manufacture more than 2 million remdesivir treatment courses by the end of this year, and several million more treatment courses in 2021.
Remdesivir is a viral RNA polymerase inhibitor which means that it interferes with the production of viral genetic material, preventing the virus from multiplying. Due to the current public health emergency, the US Food and Drug Administration (FDA) has issued an Emergency Use Authorization for remdesivir for the treatment of hospitalized patients with severe COVID-19.
“From the beginning it was clear that no one company or innovation would be able to bring an end to the COVID-19 crisis. Pfizer’s agreement with Gilead is an excellent example of members of the innovation ecosystem working together to deliver medical solutions,” Pfizer CEO Albert Bourla said. “Together, we are more powerful than alone. As one of the largest manufacturers of vaccines, biologics and sterile injectables, it is a privilege to offer our expertise and infrastructure to help fight this pandemic.”
In a separate statement, the UK’s Hikma Pharmaceuticals announced that it signed a non-exclusive supply agreement with Gilead to produce remdesivir for injection at its factory in Portugal.
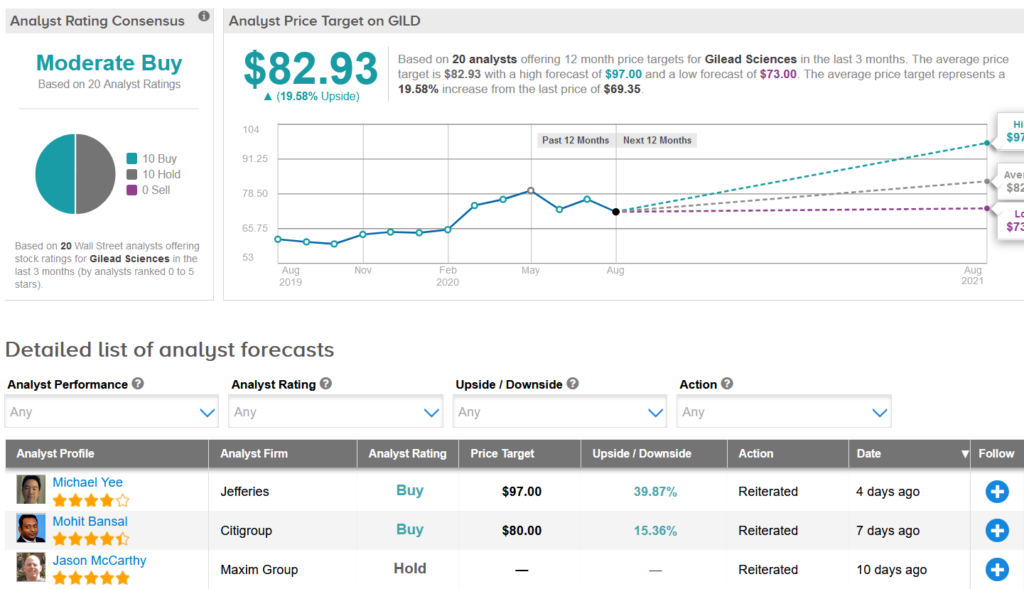
Shares in Gilead have advanced about 7% year-to-date. The $82.93 average price target implies another 20% upside potential in the shares in the coming 12 months.
Citigroup analyst Mohit Bansal earlier this month raised GILD’s price target to $80 from $78 and kept a Buy rating on the shares, as he expects Q3 remdesivir sales of $1.6 billion, supported mainly by US sales from 500,000 courses priced at $500 per vial using an 80%/20% commercial to government split.
For Q4, Bansal estimates that remdesivir sales will be driven by 625,000 courses at $390 per vial to outside of the US countries. Both calculations would add up to $3.1 billion to second-half revenue.
The rest of the Street is cautiously optimistic on the stock with a Moderate Buy analyst consensus based on 10 Buy ratings versus 10 Hold ratings. (See Gilead stock analysis on TipRanks).
Related News:
AstraZeneca Strikes First China Manufacturing Deal For Covid-19 Candidate
Amarin’s Vascepa To Take Part In Covid-19 Study In Adults With Heart Disease
Moderna Secures $400M In Deposits For Supply Of Covid-19 Vaccine Candidate









