Novavax, Inc. (NASDAQ: NVAX) disclosed that its COVID-19 vaccine, NVX-CoV2373, showed cross-reactivity against Omicron and other existing variants from a primary 2-dose regimen. NVAX also informed that responses increased after a third dose was administered at six months.
Shares of the biotechnology company gained 5% during Wednesday’s extended trade to close at $192.50.
Meanwhile, the Phase 3 trial of Novavax’s candidate in the U.S. and Mexico reflects a two to four-fold higher response in adolescents than adults against all variants, following a 2-dose series.
The company said that the single booster dose of 5 µg SARS-CoV-2 rS with 50 µg Matrix-M™ adjuvant was injected to adults about six months after the completion of their primary 2-dose vaccination series. Notably, multiple processes of analysis were used to evaluate immune responses against SARS-CoV-2 28 days after the booster dose.
President of Research and Development at Novavax, Gregory M. Glenn, said, “Given the continued evolution of the coronavirus, the development of an Omicron vaccine could be necessary. Novavax has cloned, expressed and characterized the Omicron spike protein vaccine and will soon enter the GMP-phase of production. We expect to begin clinical studies in the first quarter of 2022.”
Stock Rating
Recently, H.C. Wainwright analyst Vernon Bernardino reiterated a Buy rating and a price target of $294 (60.4% upside potential) on the stock.
Currently, the Street has a bullish outlook on the stock, with a Strong Buy consensus rating based on 3 Buys and 1 Hold. The Novavax price target of $270 implies 47.3% upside potential from current levels.
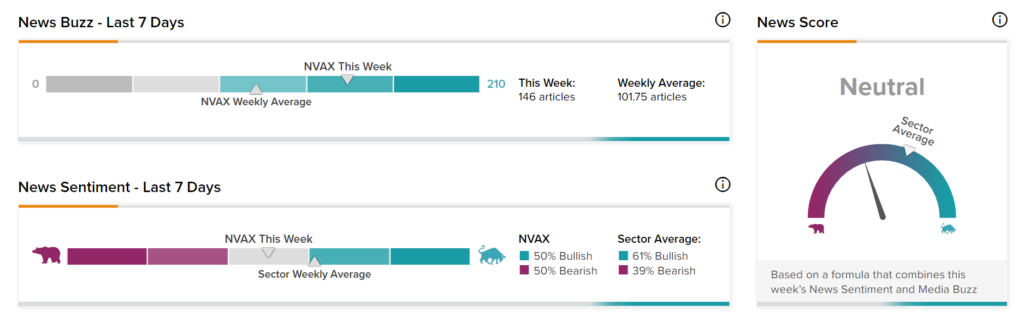
News Sentiment
News Sentiment for Novavax is Neutral, based on 146 articles over the past seven days. About half the articles have Bullish sentiment, compared to a sector average of 61%, and the remaining half have Bearish Sentiment, compared to a sector average of 39%.
Related News:
CarMax Plunges 6.7% Despite Robust Q3 Results
Regulators to Investigate Tesla’s Passenger Play Feature – Report
Ligand and GlaxoSmithKline Announce Expansion of Agreement









