Life sciences company Emergent BioSolutions Inc. has received a $23 million contract modification from Biomedical Advanced Research and Development Authority (BARDA). The company is on track with its COVID-19 vaccine manufacturing agreements and plans to use the $23 million to acquire biologics manufacturing equipment specific to Johnson & Johnson’s COVID-19 vaccine.
Emergent BioSolutions (EBS) President and Chief Executive Officer, Robert G. Kramer, said, “Emergent is actively working with the U.S. government to fulfil its policy objectives consistent with the Center for Innovation in Advanced Development and Manufacturing (CIADM) that was put in place under the Obama-Biden Administration in 2012.”
Kramer added, “This unique public-private partnership has been a key element in our ability to quickly scale up to produce COVID-19 vaccines at a current rate of more than one billion dose-equivalents annually. Emergent’s top priority continues to be the strengthening of the supply chain for Johnson & Johnson’s vitally needed COVID-19 vaccine.” (See Emergent BioSolutions stock analysis on TipRanks)
Additionally, Emergent also plans to decrease the manufacturing of AstraZeneca’s COVID-19 vaccine bulk drug substance. This decrease is in-line with a mutual agreement of the company with the U.S. government and AstraZeneca.
Currently, Emergent has nine COVID-19 CDMO partnerships with the U.S. government, pharmaceutical and biotech companies as well as non-government organizations.
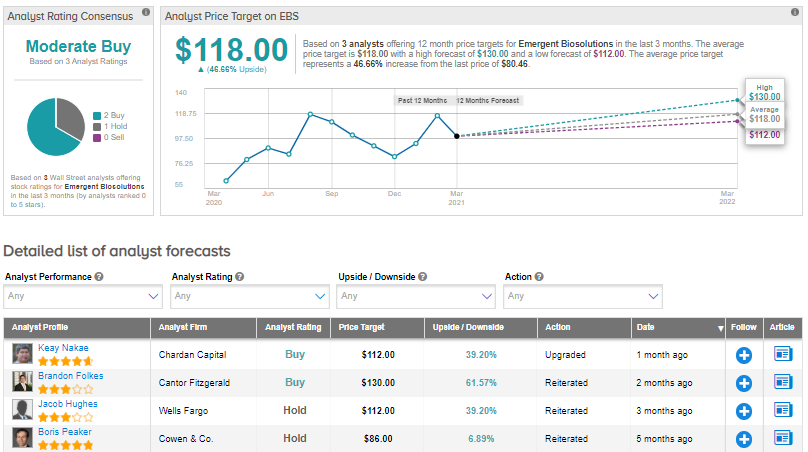
Recently, Chardan Capital analyst Keay Nakae upgraded the stock to Buy from Hold and maintained a $112 price target.
Nakae said, “2020 was a great year for Emergent. The Company’s CDMO business was a significant beneficiary of the Covid-19 pandemic, and Management did an excellent job to leverage their vaccine manufacturing capacity to capture numerous large contracts. The bulk of these agreements are ‘take or pay’ arrangements under which Emergent is being paid to reserve manufacturing capacity for these various SARS-CoV-2 vaccine initiatives.”
Turning to the rest of the Street, the stock has a Moderate Buy consensus rating alongside an average analyst price target of $118 (46.7% upside potential), based on 2 Buys and 1 Hold. Shares have gained about 48.1% over the past year.
Related News:
12 Technology Stocks Worth Watching
Abbott’s BinaxNOW COVID-19 Ag Self Test Cleared For Emergency Use In US
Acuity Brands Pops 13% After 2Q Earnings Beat, Sales Disappoint
Vipshop To Buy Back $500M In Stock; Shares Pop 9%









