Shares in ElectroCore, Inc. (ECOR) more than doubled after the U.S. Food and Drug Administration (FDA) issued an Emergency Use Authorization (EUA) for the use of its gammaCore Sapphire device to treat adult patients with COVID-19 who are experiencing exacerbation of asthma-related dyspnea and reduced airflow.
The stock surged to $2.20 in Monday’s morning market trading after the U.S. medical device maker announced that the EUA was granted for acute use of the the hand-held device at home or healthcare settings for COVID-19 patients for whom approved drug therapies are not tolerated or provide insufficient symptom relief.
The stimulation device is applying a mild electrical signal to the vagus nerve through the skin on either side of the patient’s neck. The company said that the FDA decision was based on available scientific evidence showing that it is “reasonable to believe that the device meets certain criteria for safety, performance, and labeling,” and that it may be effective in treating patients with COVID-19.
“Needless to say, we are very pleased to have received this EUA, and we intend work vigorously to make this novel therapy available to physicians treating known or suspected COVID 19 patients who are experiencing exacerbation of asthma-related breathing difficulty,” said ElectroCore CEO Dan Goldberger.
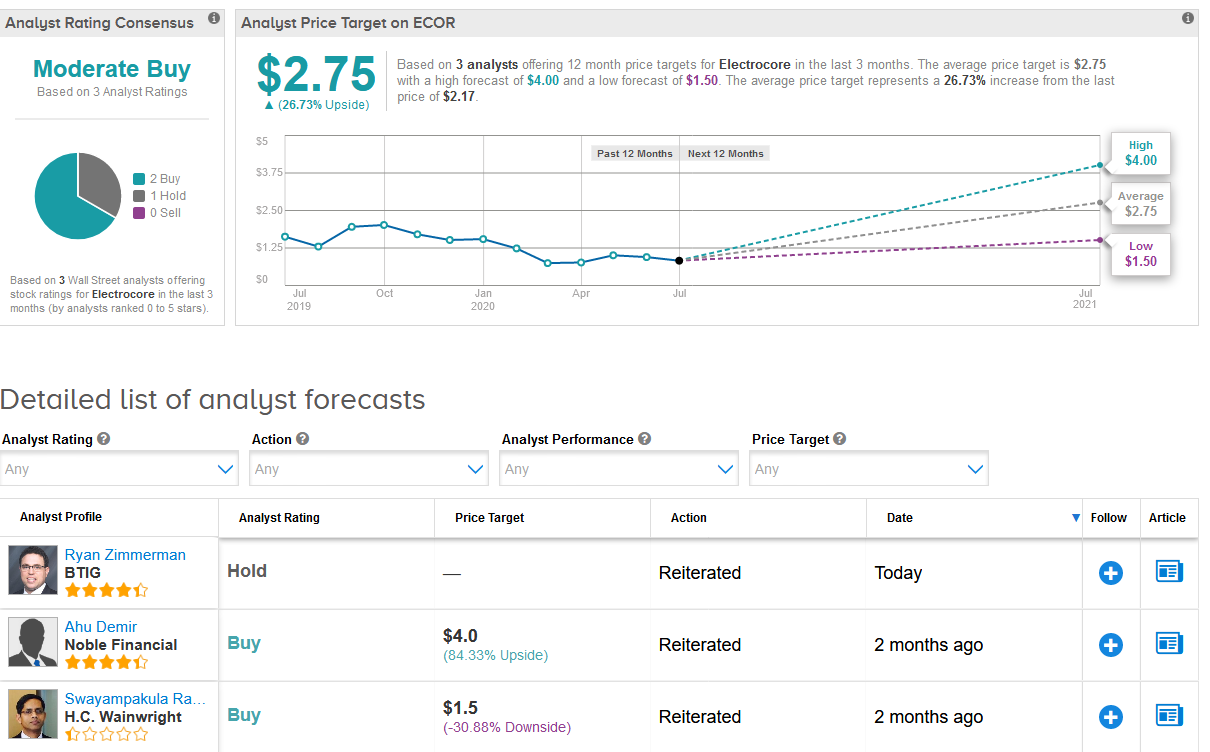
ElectroCore shares, which have plunged 47% this year, have a cautious Moderate Buy analyst consensus.
Meanwhile, Noble Financial analyst Ahu Demir reiterated a Buy rating on the stock with a $4 price target (83% upside potential), saying that the indications for gammaCore therapy represent a large commercial opportunity for the company.
“We believe the shares are poised to gain value upon revenue ramp up and the release of clinical data from patients with coronavirus and migraine (prevention),” Demir wrote in a note to investors.
Indeed, the $2.75 average analyst price target implies shares have 27% upside potential in the coming 12 months. (See ECOR stock analysis on TipRanks)
Related News:
Equillium Explodes 260% On Positive Covid-19 Results; India Approval
Gilead Reveals Covid-19 Treatment Remdesivir Reduces Mortality Risk
Moderna Inks Deal With Rovi To Supply Potential Covid-19 Vaccine Outside US