Clinical-stage biotechnology company Checkmate Pharmaceuticals Inc. reported data from its Phase 1b trial evaluating the combination of vidutolimod and pembrolizumab for the treatment of advanced anti-PD-1 refractory melanoma.
Checkmate Pharmaceuticals’ (CMPI) product candidate vidutolimod is delivered as a biologic virus-like particle. It is designed to trigger the patient’s innate immune system to target tumors in combination with other therapies.
Dr. Jason Luke, MD, Director, Cancer Immunotherapeutics Center, UPMC Hillman Cancer Center, and Associate Professor of Medicine, University of Pittsburgh School of Medicine, said, “These translational data provide new insights into the rapid pharmacodynamic responses to vidutolimod and support the conclusion that clinical responses to treatment are not associated with the previously-described predictive markers for response to PD-1 blockade such as inflamed tumors.”
In the trial, patient response rates to the drug combination were similar across baseline patient characteristics and all of the patients demonstrated the expected rapid induction of anti-Qb antibodies to the virus-like particle. (See Checkmate Pharmaceuticals stock analysis on TipRanks)
Furthermore, vidutolimod demonstrated improved response in noninflamed anti-PD-1 refractory melanoma and clinical activity was associated with serum CXCL10.
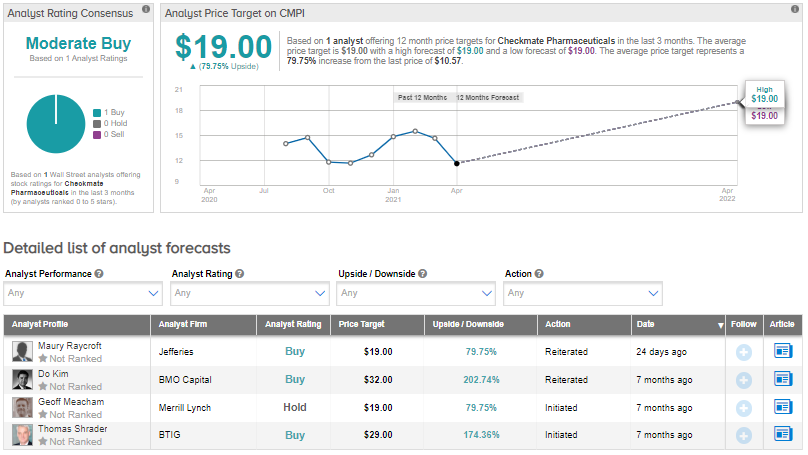
Last month, Jefferies analyst Maury Raycroft reiterated a Buy rating on the stock but lowered the price target to $19 (79.7% upside potential) from $23.
Raycroft commented, “CMPI’s lead asset ‘001 is a differentiated TLR9 agonist encapsulated in a virus-like particle in the clinic intended to augment (make cold tumors hot) existing immunotherapies to ultimately broaden efficacy in cancer.”
Shares of the healthcare company are down about 28.2% over the last month.
Related News:
Square Expands To Ireland With Early Access Programme
Levi’s 1Q Sales And EPS Beat Estimates; Street Says Buy
Provention Bio Shares Crash 40% Due To Regulatory Setback For Diabetes Drug









