Bristol Myers Squibb and bluebird bio’s Abecma received approval from the U.S. food and Drug Administration (FDA) for treating patients with relapsed multiple myeloma. Abecma is a personalized chimeric antigen receptor (CAR) T cell immunotherapy.
Bristol Myers Squibb’s (BMY) Chief Medical officer Samit Hirawat, M.D., said, “CAR T cell therapies have shown transformational potential for the treatment of hematologic malignancies, and we, with our partners at Bluebird Bio, are proud to bring the first CAR T cell therapy to appropriate triple-class exposed patients with relapsed or refractory multiple myeloma, offering the chance for durable response.”
Hirawat further added, “Bristol Myers Squibb is now the only company with two approved CAR T cell therapies with distinct targets of CD19 and BCMA.”
The therapy has a well-established safety profile and 72% of patients showed rapid, deep and durable responses in its pivotal KarMMa trial.
Notably, Bristol Myers will manufacture the drug separately for each patient using their own T cells. The two companies are developing and marketing the drug in the U.S. (See Bristol Myers Squibb stock analysis on TipRanks)
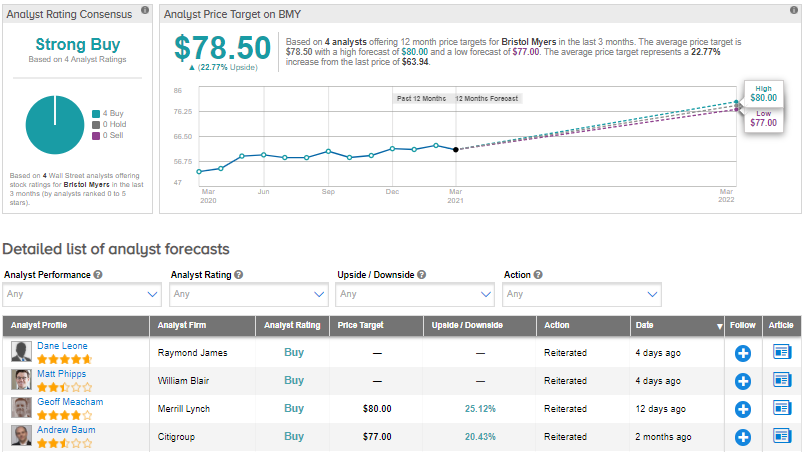
On March 17, Merrill Lynch analyst Geoff Meacham reiterated a Buy rating on the stock with a $80 price target (25.1% upside potential).
Meacham commented, “There may not be one single catalyst that will spark a re-rating, but given attractive valuation (8x vs. 13x peers) and what could be the beginning of a commercial inflection in new launches, we think the disconnect on valuation could correct pretty meaningfully in 2021.”
Turning to rest of the Street, the stock has a Strong Buy consensus rating alongside an average analyst price target of $78.5 (22.8% upside potential) based on 4 unanimous Buys. Shares have gained about 22.4% over the past year.
Related News:
MSG Entertainment Inks Deal To Buy MSG Networks; Shares Tank 10% Ametek Inks Deal To Acquire Abaco Systems For $1.35B
Synlogic Posts Lower-Than-Expected 4Q Loss; Shares Gain









