Shares of BioLineRx Ltd. skyrocketed 34% on Oct. 30 after the clinical-stage biopharma company disclosed “positive” results from its ongoing Phase 3 trial of motixafortide used in the treatment for stem cell mobilization (SCM) in multiple myeloma patients.
BioLineRx (BLRX) said that an independent Data Monitoring Committee (DMC) analyzing the study’s primary endpoint recommended that based on the statistically significant evidence favoring treatment with motixafortide, the company may already cease patient enrollment. Following the recommendation, BioLineRx said that the study enrollment is now completed with 122 patients. The study originally planned to recruit 177 patients.
Primarily focused on oncology, BioLineRX in-licenses cutting-edge compounds, develops them through clinical stages and then partners with pharmaceutical companies for further clinical development.
Full results of the motixafortide study are expected to be announced in the first half of 2021, including secondary and exploratory efficacy endpoints, as well as extended safety data, after the last patient enrolled reaches 100 days of follow-up post-transplantation.
“The compelling results of this planned interim analysis are a very significant milestone for our Company, as our SCM program is the Company’s most efficient path to registration for motixafortide,” stated BioLineRx CEO Philip Serlin. “Stem cell mobilization represents a significant unmet medical need in multiple myeloma, as between 50% and 70% of patients are poor mobilizers. We eagerly await the final results of the study, which we hope will support our goal of changing the treatment paradigm in autologous stem-cell mobilization, thus positioning motixafortide in combination with G-CSF as the new standard of care in this indication.”
The motixafortide trial was initiated in December 2017. The study is a randomized, placebo-controlled trial, evaluating the safety, tolerability and efficacy of motixafortide and G-CSF (granulocyte-colony stimulating factor), compared to placebo and G-CSF, for the mobilization of HSCs (hematopoietic stem cells) for autologous transplantation in multiple myeloma patients.
The primary objective of the study is to demonstrate that only one dose of motixafortide on top of G-CSF is superior to G-CSF alone in the ability to mobilize cells in up to two apheresis sessions. Secondary objectives include time to engraftment of neutrophils and platelets and durability of engraftment, as well as other efficacy and safety parameters.
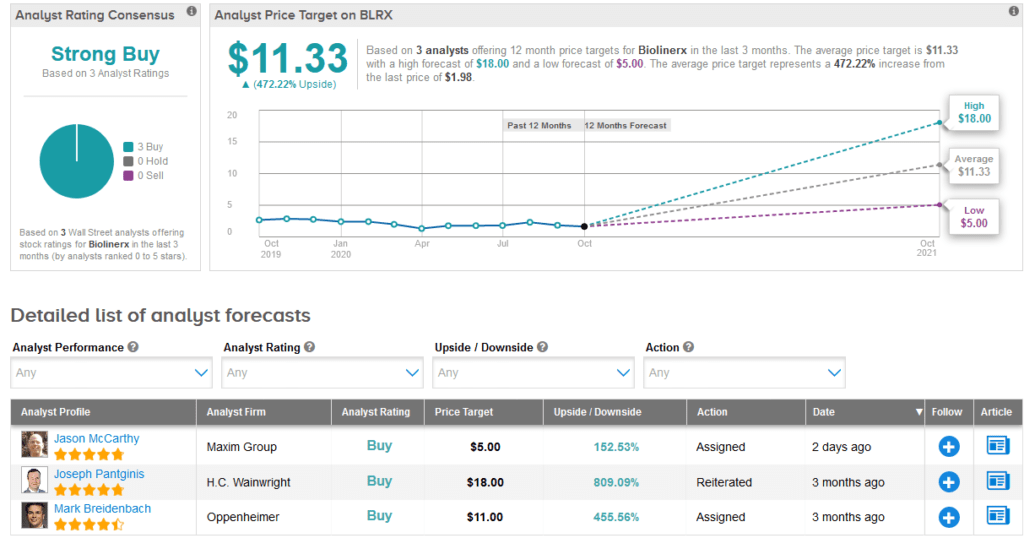
BLRX shares are down 11% from the start of the year and the stock scores a bullish Strong Buy analyst consensus backed up by 3 recent unanimous Buy ratings. Looking ahead, the $11.33 average price target, indicates investors could be reaping a stellar 472% gain in the coming 12 months.
Among the three analysts taking a bullish approach, Maxim Group’s Jason McCarthy on Oct. 30 reiterated his Buy rating on the stock with a $5 price target.
“It is important to consider that motixafortide has wide potential applicability beyond just multiple myeloma, which could make it especially attractive for a partner with the resources to fund expansion,” McCarthy wrote in a note to investors. “Expansion of motixafortide to other hematologic indications for stem cell mobilization (such as non-hodgkins lymphoma or sickle cell) could greatly increase market size with just an incremental increase in associated operating expenses (the stem cell transplantation market is concentrated, and many of the transplant centers handle multiple indications), unlocking additional value for investors.” (See BioLineRx stock analysis on TipRanks)
Related News:
AstraZeneca Sells Rights To Hypertension Drugs For $400M
Moderna Scores $1.1B From Covid-19 Vaccine Deposits; Shares Rise 3%
Opko Health Tanks 10% Despite Solid Q3 Covid Testing Revenue









