Biogen Inc. (BIIB) announced that it has finally submitted the Biologics License Application (BLA) seeking approval by the Food and Drug Administration (FDA) for aducanumab, its investigational treatment for Alzheimer’s disease.
If approved, aducanumab would become the first therapy to reduce the clinical decline of Alzheimer’s disease, the company said. The FDA application submission had been delayed since early 2020. Aducanumab is a human monoclonal antibody designed to treat early Alzheimer’s disease.
Biogen said that the completed submission followed ongoing collaboration with the FDA and includes clinical data from the Phase 3 studies, as well as the Phase 1b study.
“Alzheimer’s disease remains one of the greatest public health challenges of our time,” said Biogen CEO Michel Vounatsos. “The aducanumab BLA is the first filing for FDA approval of a treatment that addresses the clinical decline associated with this devastating condition, as well as the pathology of the disease.”
Biogen reported that clinical studies showed that patients who received aducanumab experienced significant slowing of decline on measures of cognition and function such as memory, orientation and language. Patients also experienced slowing of decline on activities of daily living including conducting personal finances, performing household chores, such as cleaning, shopping and doing laundry, and independently traveling out of the home.
The FDA now has up to 60 days to decide whether to accept the application for review. As part of the submission, Biogen requested to get priority review.
The stock rose 4.4% on the news and closed at $280.19 on Wednesday trimming this year’s decline to 5.6%. Meanwhile some analysts are skeptical about the likelihood of the drug approval. Although, Goldman Sachs analyst Terence Flynn called Biogen’s submission “an important first step and incremental positive,” he seeks risk to its approval.
“The central question is how the FDA will interpret the aducanumab Phase 3 data given a single positive trial, weighed against the unmet need in Alzheimer’s”, Flynn wrote in a note to investors adding that he sees a 20% probability of success.
The analyst maintained a Hold rating on the stock with a $300 price target (7.1% upside potential).
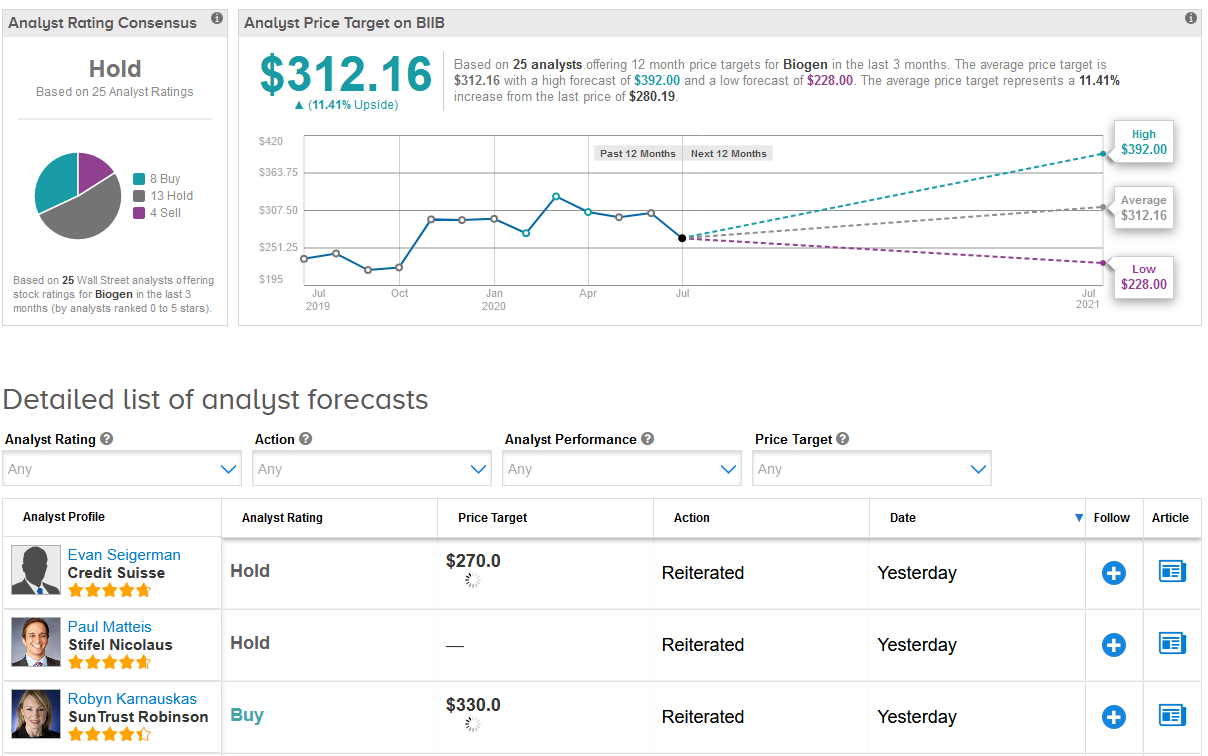
In line with Flynn’s outlook, Wall Street analysts have a Hold consensus on Biogen with a $312.16 average price target (11% upside potential). In the last three months, the stock has received 13 Hold ratings and 4 Sell ratings versus 8 Buy ratings. (See BIIB stock analysis on TipRanks).
Related News:
Novavax Spikes 42% Pre-Market On $1.6B U.S. Funding For Covid-19 Candidate
Corvus Shoots Up 115% On Start Of Novel Immunotherapy Study In Covid-19 Patients
GenMark Soaring In Pre-Market On 118% Revenue Explosion