Shares in AstraZeneca (AZN) dropped 3.5% even after early-stage human trial data from its Covid-19 vaccine candidate showed “robust” immune responses.
The stock declined to $58.96 in afternoon market trading. Interim data results from the ongoing Phase I/II trial of the potential coronavirus vaccine, also known as AZD1222, which AstraZeneca is developing with Oxford University, showed “strong” antibody and immune T-cell responses.
“While there is more work to be done, today’s data increases our confidence that the vaccine will work and allows us to continue our plans to manufacture the vaccine at scale for broad and equitable access around the world,” said Mene Pangalos, Executive VP of BioPharmaceuticals R&D.
The results demonstrated that a single dose of AZD1222 resulted in a four-fold increase in antibodies to the SARS-CoV-2 virus spike protein in 95% of participants one month after injection. In all participants, a T-cell response was induced, peaking by day 14, and maintained two months after injection.
Neutralising activity against SARS-CoV-2 was seen in 91% of participants one month after vaccination and in 100% of participants who received a second dose. The levels of neutralising antibodies seen in participants receiving either one or two doses were in a similar range to those seen in convalescent COVID-19 patients.
Late-stage Phase II/III trials are currently underway in the U.K., Brazil and South Africa and are due to start in the U.S., AstraZeneca said.
The British drugmaker has in recent weeks signed supply chain agreements for the capacity to produce 2 billion doses of its vaccine candidate, should it prove to be successful. It has inked supply deals with the U.S. and European Union countries.
AZD1222 is one of several candidates supported by Operation Warp Speed (OWS), the U.S. government program to accelerate the development, manufacturing, and distribution of COVID-19 vaccines available for Americans by Jan. 2021.
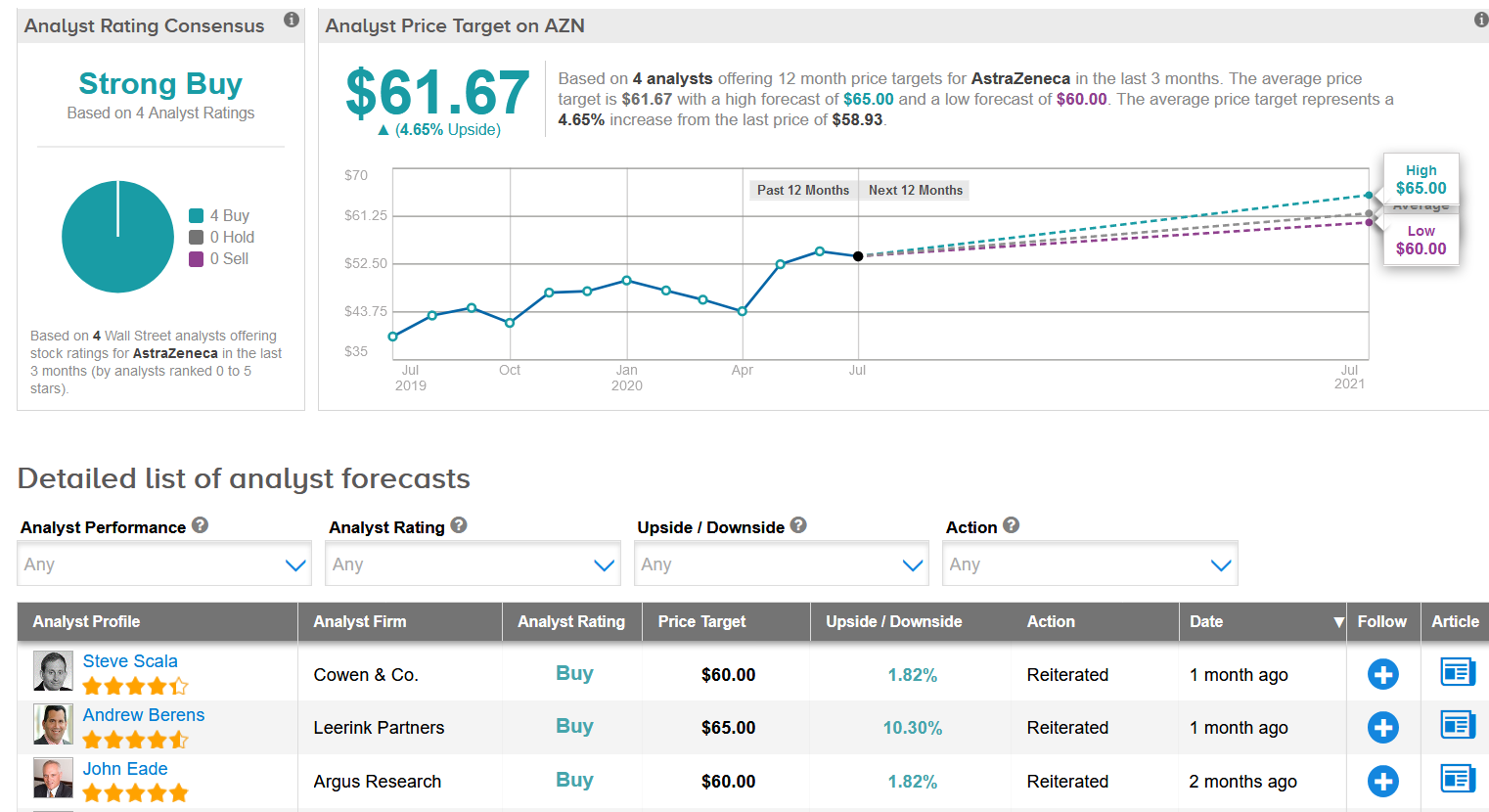
Shares have jumped 10% in the past five days in anticipation of the trial results and are now up more than 18% year-to-date. In light of the recent rally, the $61.67 average analyst price target puts the upside potential at a more modest 4.7% in the coming 12 months. (See AstraZeneca stock analysis on TipRanks)
Overall, the stock scores a Strong Buy consensus from the analyst community based on 4 unanimous Buy ratings.
Related News:
Pfizer, BioNTech Ink UK Supply Deal For 30M Covid-19 Vaccine Doses
Moderna Soars 16% As Covid-19 Vaccine Shows Strong Immune Response
GSK Buys 10% Stake In Germany’s CureVac To Develop mRNA Vaccines









