Analysts from Robert W. Baird and Roth Capital explain why they are bullish on rival drug-maker Sarepta Therapeutics Inc (NASDAQ:SRPT) and biopharmaceutical firm Celator Pharmaceuticals Inc (NASDAQ:CPXX) following an open letter to the FDA regarding SRPT’s eteplirsen in DMD, and CPXX’s quarterly update.
Sarepta Therapeutics Inc
Analyst Brian Skorney of Baird weighed in on SRPT after 36 doctors issued a 3 page letter to the FDA urging the approval of drug eteplirsen, an experimental drug designed to treat aspects of Duchenne muscular dystrophy. The FDA’s Peripheral and Central Nervous System Drugs Advisory panel is set to meet on April 25 to discuss the NDA of eteplirsen. In the letter, the 36 DMD experts both urged the FDA to accelerate approval the drug and also combated its previous claims of insufficient data on the drug’s effectiveness.
The analyst reiterated an Outperform rating and $47 price target. He states, “We believe this is a very significant asset for Sarepta going into next month’s panel. It essentially creates a we (36 doctors and scientists dedicated to the study and treatment of DMD) know better than Farkas (the FDA’s team leader on the eteplirsen review) dynamic. Even the most skeptical person is going to find it hard to argue that a regulators interpretation of a data set is more relevant than the interpretation of those with the most experience (over 5,000 DMD patients).”
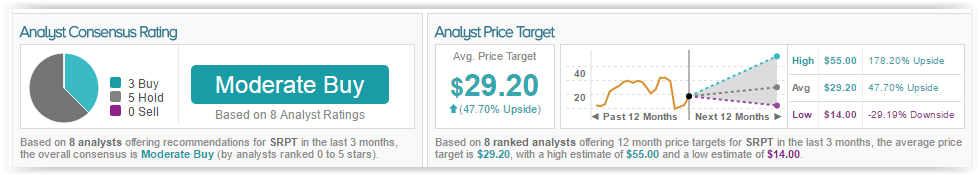
Brian Skorney has a 42% success rate recommending stocks with an average loss of (2.1%) per recommendation, according to TipRanks. Out of the 8 analysts who rated the company in the past 3 months, 3 gave a Buy rating while 5 remain on the sidelines. The average 12-month price target for the stock is $29.30, marking a 48% upside from current levels.
Celator Pharmaceuticals Inc
Roth analyst Joseph Pantginis expressed his views on CPXX after the company released Q4:15 earnings, posting in line EPS of ($.13) per share, compared to consensus of ($0.12) per share, and cash of ($9.8) million in 1Q16 resulting from an outstanding ATM facility. The analyst chose to focus more on recently released phase 3 data for Vyxeos, a liposome injection to treat high-risk acute myeloid Leukemia. The trial showed significant improvement in overall survival rates, meeting its primary endpoint. Those treated with Vyxeos displayed a median survival time of 9.56 months vs the 5.95 months in patients receiving the “7+3” regimen, the disease’s traditional treatment.
The drug represents the first in decades to display survival improvements in induction therapy for AML. As a result of this breakthrough data, the analyst believes the company will file an NDA and MAA in 3Q16 and 1Q17, respectively. He believes the drug will be popular with doctors, explaining, “It is our view that Vyxeos will supplant the standard of care 7+3 therapy and adoption rates should be high since physicians do not need to learn anything new and it should be easy to explain to patients.”
The analyst also comments on a potential partnership to market the drug. He explains, “Additionally, now that the program has been de-risked, we believe there is high potential for CPXX to deliver an ex-U.S. partner,” exploring its cash options “in order to build out its commercial infrastructure.”
The analyst reiterates his Buy rating and $22 price target on the company. He states, “Vyxeos now represents the first meaningful improvement to treating AML in decades and look to both expansion into additional AML indications as well as further visibility from the CombiPlex platform.” Joseph Pantginis has a 32% success rate recommending stocks with an average loss of (7.5%) per recommendation.